industrial vs. photochemical smog
industrial vs. photochemical smog
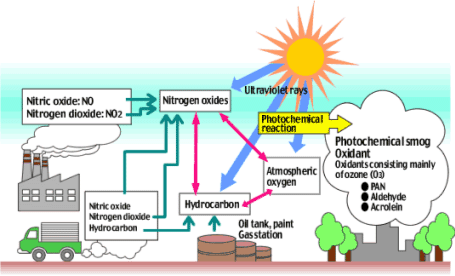
Industrial and photochemical smog are both forms of air pollution that appear to be a yellow-brown haze, but they differ in both their formation and composition. Industrial smog is formed when sulfur dioxide and particulates, released by the combustion of coal, combine with water in fog to produce an acidic haze . One of the worst cases of industrial smog was the Great Smog of 1952 in London, where the smog was so thick it killed thousands of people and purportedly caused cows to suffocate ("The Great Smog of 1952," 2015). Photochemical smog is formed when a chemical reaction between nitrogen oxides and VOCs due to sunlight forms dangerous secondary pollutants, such as ozone and peroxyacetyl nitrate. Cities with high population density, such as Los Angeles, New York, Sydney, and Vancouver often have significant amounts of photochemical smog. Both forms of smog cause breathing problems and skin irritation, but photochemical smog can damage lung tissue and the immune system, and industrial smog can kill people with its sulfuric acid (Harvey, n.d.).